< Previous | Table of Contents | Next >
Central to the SOPs Regime is the concept of SMSE. Both the RMA and the SMRC must base their decisions on SMSE as defined in the s5AB of the VEA which states:
- Information about a particular kind of injury, disease or death is taken to be sound medical-scientific evidence if:
- the information:
- is consistent with material relating to medical science that has been published in a medical or scientific publication and has been, in the opinion of the Repatriation Medical Authority, subjected to a peer review process; or
- in accordance with generally accepted medical practice, would serve as the basis for the diagnosis and management of a medical condition; andin the case of information about how that kind of injury, disease or death may be caused—meets the applicable criteria for assessing causation currently applied in the field of epidemiology.
Evaluation of sound medical-scientific evidence
The RMA can only make a SOP about a particular kind of injury, disease or death where there is sufficient SMSE to justify the making of the SOP. The SMRC when reviewing a decision made by the RMA must base its decision on the SMSE available to and obtained by the RMA6.
The RMA and the SMRC are required to assess material against certain epidemiological criteria. The criteria set out below are not exhaustive and may not be relevant in all cases. They are a guide to the material that is acceptable to the RMA and the SMRC.
Quality of evidence
In assessing studies, both the RMA and the SMRC look for SMSE that:
- is well-designed;
- provides enough information;
- is not merely hypothesis generating exercises from large databases;
- has adequate outcome measurements; and
- has no other faults in the methodology
Assessing Combined Epidemiological Evidence
The RMA and the SMRC examine the body of evidence against these criteria:
- strength of association;
- consistency;
- specificity;
- temporality;
- biological gradient;
- plausibility;
- experimental evidence; and
- analogy.
Websites
Website articles are not SMSE if they are opinion based, not subject to peer review, or do not meet the applicable criteria for assessing causation currently applied in the field of epidemiology.
Animal Studies
Animal studies may sometimes support the biological plausibility of an association. However, results from animal studies may not be generalisable to humans and are at best used as initial research that may indicate a need for further studies on human subjects or to demonstrate possible biological mechanisms.
Laboratory Studies
Laboratory-based studies of human cells are used in medical research for exploring potential pathological mechanisms, such as examining inflammatory responses to toxins. Processes occurring at the cellular level can be misleading as many other processes contribute to human health effects. While such studies may demonstrate biological mechanisms or generate further research, only some would lead to further discoveries, and they can often produce a range of conflicting results.
Such studies can be material that epidemiologists would consider appropriate to take into account, but the weight attached to their results when considering causes of diseases varies, and is generally relatively low compared to human studies.
The RMA’s process for sourcing evidence for its briefing papers follows standard practices for systematic reviews.
The following diagram summarises the process of SOP determination.
Figure 1 Determination of Statements of Principles
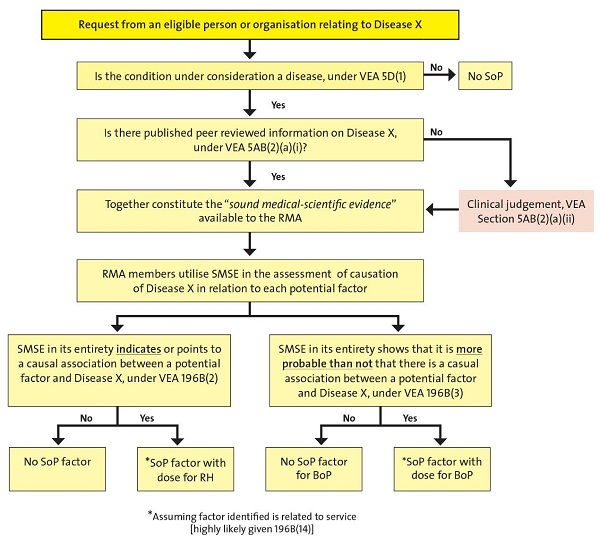
The RMA writes the SOPs, but does not consider individual claims for pension, nor specify the ways in which factors in a SOP may be related to service.
Investigations and reviews are undertaken on request from eligible parties7, at direction by the SMRC, or on the RMA’s own initiative. The Legislative Instruments Act 2003 (LIA) requires the RMA to review and reissue each SOP at least every 10 years, failing which a SOP ceases to have legal effect.
< Previous | Table of Contents | Next >
6The SMRC does not conduct new literature searches and cannot rely upon ‘new’ information that has not been considered by the RMA.
7S196E(1) of the VEA specifies the persons and organisations eligible to request an investigation or review.